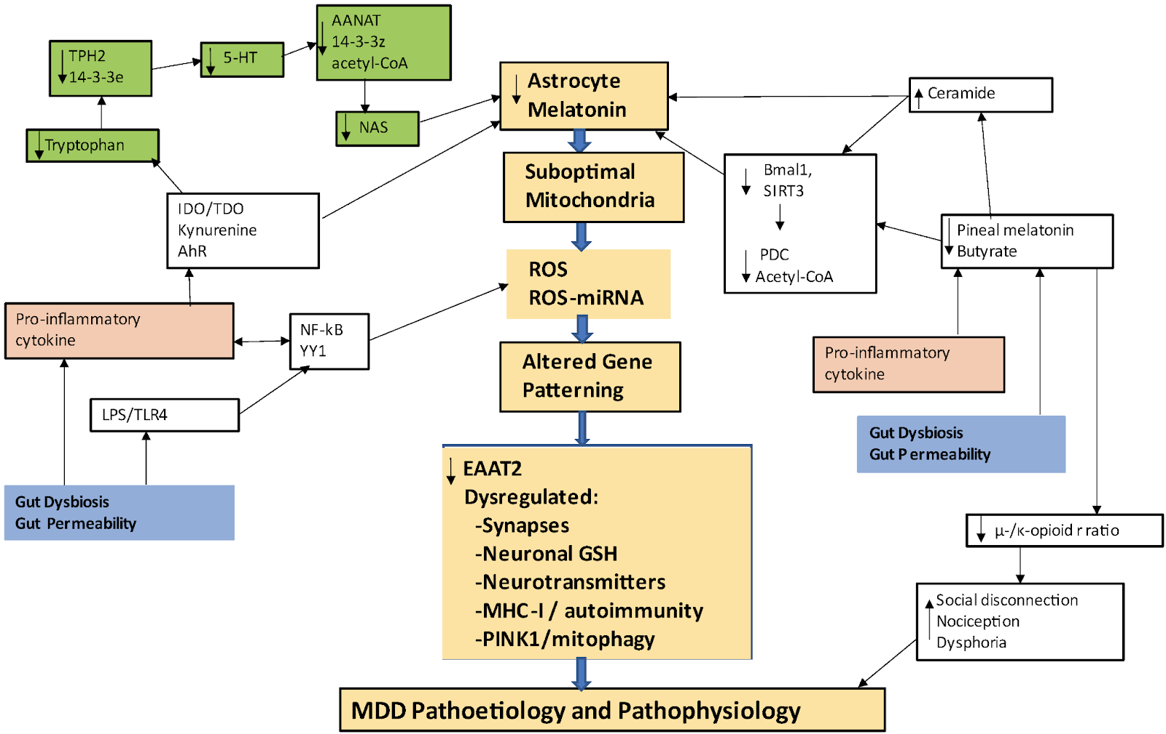
Mental health disorders, such as anxiety, depression, and post-traumatic stress disorder (PTSD), affect millions of lives worldwide. In recent years, regenerative medicine has emerged as a promising frontier in the battle against these disorders. In this blog post, we delve into the molecular foundations of mental health issues and explore how regenerative medicine, particularly treatments involving the vagus nerve, can offer new hope. An overview of the pathogenesis of depression is shown below. This schematic was taken from this paper by George Anderson.
Molecular Mechanisms of Mental Health Disorders
Neuroinflammation
The immune system, the body’s natural defense mechanism against pathogens, can become detrimental when excessively activated, particularly within the brain, where neuronal regeneration is constrained. Notably, leukocytes are almost absent from the healthy central nervous system (CNS) parenchyma. Instead, resident immune cells, such as microglia and astrocytes, play a central role. They promptly and non-selectively eliminate pathogens and foreign entities through inflammation.
However, when triggered by stress, trauma, pathology, or infection, microglia and astrocytes initiate a cascade of pro-inflammatory molecules, including reactive oxygen species and cytokines. These signalling agents attract myeloid and lymphoid cells into the brain. This activation also leads to the recruitment of adaptive immune cells tailored to the specific pathological agent, such as B and T lymphocytes.
At the core of this intricate process lies the bidirectional communication between the central and peripheral immune systems, with the vagus nerve serving as a crucial connection. When functioning optimally, the vagus nerve helps to maintain a delicate balance in the immune response, preventing excessive inflammation. However, in cases where the vagus nerve’s regulatory mechanisms become compromised—often due to stress, trauma, or chronic inflammation—it can lead to dysregulation of the immune response within the CNS. This dysregulation can trigger neuroinflammation, setting off a cascade of events that contribute to the pathogenesis of mental health conditions.
Recent research has illuminated the central role of neuroinflammation in mood disorders and various mental health conditions. Conditions such as depression and anxiety have been linked to the dysregulation of the CNS immune system. Pro-inflammatory cytokines, including IL-1β and IL-6, have been found to be elevated in individuals with depression, and drugs targeting neuroinflammation, such as COX-2 inhibitors, are now under consideration as potential treatments for psychiatric diseases.
Neurotransmitter Imbalances
Neurotransmitters are signalling molecules in the brain. Several key neurotransmitters are involved in neuromodulation, regulating brain activity globally. These include key molecules such as serotonin, dopamine, and norepinephrine, and they help regulate mood, emotion, and cognitive function. It is thought that one contributing factor to psychiatric illness is an imbalance in these neurotransmitters, termed the monoamine hypothesis. In fact, drugs developed to treat mental health conditions like depression, anxiety, or attention-deficit hyperactivity disorder (ADHD) specifically target pathways to alter the levels of these neurotransmitters in the brain.
Gut-Brain Axis
The gut houses microorganisms referred to as the gut microbiota, which generate neurotransmitters and inflammatory compounds capable of affecting both brain function and mood regulation. Dysbiosis, an imbalance in this microbiota, may result in the creation of pro-inflammatory molecules that can circulate in the bloodstream and impact the brain, potentially leading to neuroinflammation. Moreover, the gut microbiota regulates the production, transportation, and functioning of neurotransmitters like serotonin or dopamine. A schematic is shown below.