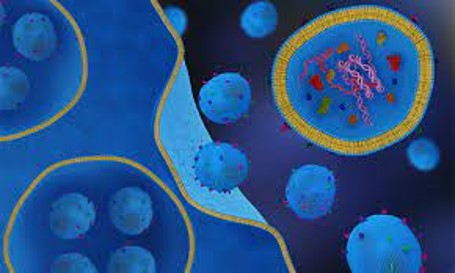
Exosomes are nanosized extracellular vesicles ranging from 30 to 150 nanometers in diameter, secreted by almost all cell types, including immune cells, stem cells, and cancer cells. First discovered in the 1980s, exosomes were initially considered cellular waste disposal mechanisms. However, subsequent research revealed their crucial involvement in intercellular communication; and recent research has suggested that exosomes might critical to treating presently incurable diseases.
Exosomes are small extracellular vesicles originating from cells when an intermediate endocytic compartment called the multivesicular body (MVB) fuses with the plasma membrane. This fusion process releases intraluminal vesicles (ILVs) into the extracellular environment, and these released vesicles are what we commonly refer to as exosomes.
Roles in Intercellular Communication
Exosomes act as messengers between cells, facilitating the exchange of information and influencing recipient cell behavior. They can be taken up by neighboring or distant cells through endocytosis, phagocytosis, or membrane fusion. Once internalized, exosomal cargo can modify the recipient cell’s gene expression, signal transduction pathways, and immune responses. Exosomes have been found to cross the blood-brain barrier, facilitating intercellular signaling in the brain.
Exosomes in Neurological Disease
The nervous system is a highly intricate system of communication, facilitating rapid transmission of electrical signals between neurons at speeds of up to 120 meters per second, allowing for near-instantaneous connections within the human brain. Given that exosomes are crucial mediators of intercellular communication, they play an essential role in the nervous system and may hold the key to treating neurological diseases.
Exosomes in Alzheimer’s Disease
Alzheimer’s Disease (AD) is the most common form of dementia and a leading cause of disability worldwide. AD is characterized by insidious-onset brain damage that is closely correlated with the formation of amyloid-beta (Aβ) plaques in the brain. Aβ plaques are abnormal protein deposits that accumulate between nerve cells (neurons) in the brain, disrupting normal cellular function and communication. At present, there is no cure for AD.
Exosomes are critical in the pathogenesis of AD. Exosomes can spread pathological proteins like misfolded Aβ, facilitating their spread and amplification throughout the brain in a process known as “seeding.”
Interestingly, exosomes are also being investigated as potential therapeutic agents in treating AD. Exosomes can transport RNA, remain stable in bodily fluids, and cross the blood-brain barrier. Through these mechanisms, exosomes may exhibit neuroprotective effects in AD by delivering disease-modifying drugs, clearing toxic waste, and mitigating glutamate excitotoxicity.
Exosomes in Parkinson’s Disease
Parkinson’s Disease (PD) is the second most common neurodegenerative disease. The pathogenesis of Parkinson’s disease is the gradual loss of dopamine-producing neurons in the substantia nigra. Dopamine is a crucial neurotransmitter involved in regulating movement, and its deficiency leads to the characteristic motor symptoms of PD such as shuffling gait, bradykinesia, and resting tremor. Clinical treatment improves symptoms but cannot protect these dopamine neurons effectively.
The pathophysiology of PD, like AD, relies on misfolded protein aggregates in the brain. In PD, this protein is called alpha-synuclein, and aggregates are known as Lewy bodies. As in AD, exosomes play a pathologic role by facilitating the spread of alpha-synuclein throughout the brain. However, recent research also implicates exosomes in treating PD. A recent Nature paper studied exosomes secreted by human umbilical cord mesenchymal stem cells and found that in vivo, these exosomes successfully reached the substantia nigra and reduced the loss of dopaminergic neurons.
Exosomes in Traumatic Brain Injury
Traumatic Brain Injury (TBI) is another common neurologic condition that also lacks adequate disease-modifying treatment. In TBI, a traumatic force to the skull is transmitted to the brain, resulting in primary physical damage. This is followed by secondary damage (e.g. electrochemical changes, ischemia, inflammation) that further damages brain tissue. Traumatic brain injury (TBI) results in severe damage to neurons and often leads to fatal consequences. Despite extensive efforts, no clinically successful treatment strategy for repairing brain injuries or neural damage has been reported. One major challenge in TBI treatment is delivering drugs effectively through the blood-brain barrier. To address this, researchers are exploring exosomes. Research is revealing that exosomes can decrease neuroinflammation post-TBI and can help promote nervous system repair post-TBI.
Patient Story
At Eterna Health, we treated a patient who had a stroke four years prior, which resulted in the loss of his left lower extremity. After IV exosome treatment, he felt a sensation in that same limb a day later, and he could move it a week later. After a week, he was walking again!
Conclusion
Exosomes represent a fascinating and multifaceted aspect of intercellular communication. Their unique composition and role as cellular messengers make them attractive targets for further research and potential clinical applications, especially in neurologic diseases. As our knowledge of exosome biology deepens, these tiny vesicles are poised to become increasingly prevalent across diverse disease contexts.